Proiect PN-III-P1-1.2-PCCDI-2017-0769 (Contract 64PCCDI/2018) ONCORAD
Dezvoltarea de radiofarmaceutice si tehnici nucleare in oncologie pentru imagistica si tratament personalizat la nivel molecular
Buget total: 5287500 RON
Institutia Coordonatoare: Institutul National de Cercetare si Dezvoltare pentru Fizica si Inginerie Nucleara “Horia Hulubei” (IFIN-HH), Director Proiect: Dr. Mihai Ciubotaru
Titlul proiectului component P1: Dezvoltarea de anticorpi anti markeri specifici evolutivi pe linia mieloida pentru radiodetectia si tratamentul leucemiilor mieloide in stadiile incipiente
Buget proiect P1: 1595414 RON (Buget INCD-VB: 423.011 RON)
Responsabil proiect P1: Dr. Mihai Ciubotaru
Institutiile implicate in P1:
Institutul National de Cercetare si Dezvoltare pentru Fizica si Inginerie Nucleara “Horia Hulubei” (IFIN-HH);
Institutul de Biochimie al Academiei Romane (IBAR);
Institutul National de Cercetare Medico-Militara “Cantacuzino” (IC);
Institutul National de Cercetare-Dezvoltare in Domeniul Patologiei si Stiintelor Biomedicale “Victor Babes” (IVB)
Echipa IVB implicata in P1: Valeriu Cismasiu (responsabil), Sevinci Pop, Gisela Gaina, Cristina Niculite, Stefania Rogozea/Alexandra Popa, Ioana Lambrescu, Victor Ionescu
Abstract :
Proiectul ONCORAD si platforma tehnologica de radiobiologie asociata sunt construite pe fundamentele medicinei translationale si ale medicinei personalizate, si au ca obiectiv principal dezoltarea de produse radiofarmaceutice tintite si tehnici medicale nucleare inovative pentru diagnostic imagistic si tratament imbunatatit in cancer. Proiectul ONCORAD se bazeaza pe competenta unica in domeniul radioizotopilor si a radiofarmaceuticelor de la Institutul coordinator (IFIN-HH), complementand-o cu expertiza din institutii partenere cu activitate prodigioasa de cercetare in domeniul biomedical (IVB, IC si IBAR), clinici (Institutul Clinic Fundeni, Bucuresti), ca si din domeniul nanotehnologiilor/nanomedicinei (INCD pentru Tehnici Izotopice si Moleculare, Cluj-Napoca). Aceasta mobilizare multidisciplinara se concretizeaza in patru proiecte de cercetare interconectate, cu obiective in domeniul medicinei nucleare oncologice. In plus, proiectul ONCORAD va sprijini dezvoltarea institutiilor din consortiu prin angajarea de tineri cercetatori si instruirea acestora in domenii stiintifice si tehnologice de varf, dezvoltarea infrastructurii de cercetare, transferul de cunostinte, tehnologii si bune practici intre partenerii din consortiu si catre alte parti interesate. Proiectul creaza cadrul de colaborare interdisciplinara intre institutiile partenere, constituind astfel premiza pentru participarea viitoare a consortiului in proiecte, diversificarea ofertelor de cercetare, transfer tehnologic pentru aplicatii clinice.
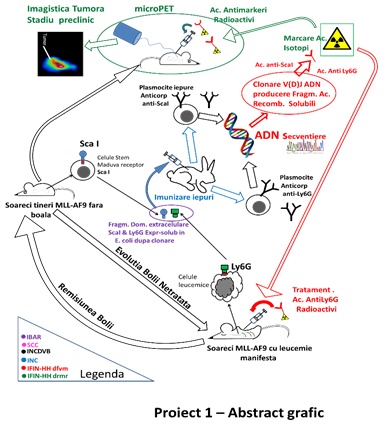
Obiectivul stiintific principal al Proiectului 1 a fost obtinerea unor anticorpi si fragmente monocatenare Fab imune impotriva unor markeri evolutivi de suprafata atat pentru stadiile timpurii stem Sca1 (stem cells antigen 1) cat si de diferentiere mieloida (Ly6C) prezenti pe suprafata celulelor de tip mononuclear neoplazic in stadiile incipiente ale leucemiilor mieloide. Aceste fragmente monocatenare vor fi marcate cu izotopi emitatori de pozitroni (Zr-89, Cu-64) in scopul folosirii lor ca biomarkeri. Detectia in vitro a celulelor leucemice se va realiza in culturi prin tehnici nucleare, iar in vivo prin micro PET-CT (tomografie prin emisie de pozitroni cuplata cu tomografie computerizata, cu rezolutie sub-milimetrica). Cuplarea fragmentelor imune monocatenare cu radionuclizi terapeutici prin tehnici complementare pot conduce la anticorpi si cu rol terapeutic, a caror eficienta in oprirea cresterii tumorale va fi testata prin tehnici in vitro si in vivo. Radionuclizii terapeutici care vor fi testati in cadrul proiectului sunt emitatori de radiatie cu transfer de energie mare (potential toxic ridicat), At-211/Ac-225 si/sau Cu-64/67.
Echipa de la IVB a dezvoltat modelul in vivo pentru leucemia mieloida acuta, a analizat si caracterizat fenotipic celulele tumorale. Ulterior, grupul de cercetare de la INCD-VB a testat noii anticorpi anti-Sca1 si anti-Ly6C, in vederea selectiei clonelor cele mai eficiente pentru marcarea radioactiva.
Rezultatele echipei de la IVB
Pentru generarea modelului de leucemie mieloida acuta a fost utilizata linia de soareci STOCK Kmt2atm2(MLLT3)Thr/KsyJ (The Jackson Laboratory, # 009079). Transgena care mimeaza translocatia cromozomala t(9;11)(p22;q23) este inserata in locusul genei Kmt2a. Translocatia determina anularea sintezei proteinei murine MLL, care este inlocuita cu proteina de fuziune MLL-AF9 (MA9). Selectia animalelor purtatoare ale mutatiei s-a realizat prin genotiparea cu PCR. Mutatia determina o instabilitate epigenetica a celulelor hematopoietice care se finalizeaza cu transformarea oncogenica completa si formarea celulei leucemice stem. Astfel, atat celulele stem hematopoietice (HSC) cat si celulele precursoare ale granulocitelor si monocitelor (GMP), purtatoare ale mutatiei MA9 sunt considerate celule pre-leucemice (p-HSC, p-GMP). Aparitia si evolutia leucemiei mieloide a fost urmarita prin monitorizarea splinei si a starii generale a animalelor. In figura 1 este prezentata curba de supravietuire a animalelor precum si dimensiunile marite ale splinei (semn al infiltratiilor celulelor tumorale).
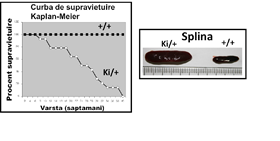
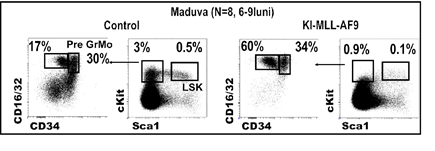
Durata medie de transformare oncogena este de 25 saptamani, asa cum rezulta din analiza Kaplan-Meier. In figura 2 se observa ca mutatia MA9 favorizeaza mielopoieza si cresterea populatiilor de granulocite si monocite (marcate cu CD45 si CD11b). Rezultatele demonstreaza ca transformarea oncogena este insotita de o reducere a populatiei stem (pre)leucemica p-HSC (denumita LSK in figura 2). Al doilea fenomen asociat cu dezvoltarea leucemiei in soarecii MA9 este cresterea populatiei precursorilor granulo-monocitari (pre)leucemici p-GMP (poarta preGrMo, in figura 2). Rezultatele sunt in concordanta cu un studiu publicat conform caruia diferentierea mieloida este necesara pentru transformarea tumorala si aparitia leucemiei mieloide acute (Ye M et al. 2015, Cell Stem Cell 17:611-623).
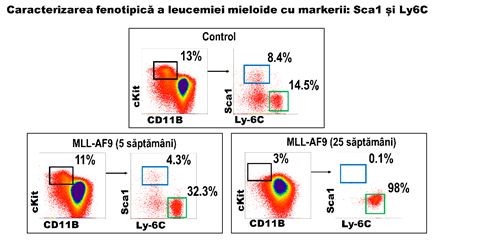
Rezultatele prezentate arata ca evolutia tumorala este asociata cu reducerea populatiei stem si, simultan, cu o crestere a progenitorilor granulo-monocitari. Prin urmare identificarea unor markeri specifici acestor doua categorii de celule va permite monitorizarea in vivo a transformarii oncogenice. Analiza a condus la identificarea a doi markeri adecvati: Sca-1 si Ly6C. In figura 3 sunt prezentate rezultalele semnificative unui set de 4 determinari independente. Se observa reducerea progenitorilor hematopoietici (cKit+, dreptunghi negru) in soarecii MA9 inca de la varsta de 5 saptamani, ceea ce indica a reducerea populatiilor stem (marcate cu Sca1). In concordanta cu acest aspect, se observa reducerea populatiei pozitive pentru Sca1 (fig 3, dreptunghi albastru) la 5 saptamani si disparitia acestor celule in soarecii cu leucemie acuta mieloida (MLLAF9, 25 saptamani). In schimb, populatia cKit pozitiv Ly6C pozitiv (figura 3, dreptunghi verde) se mentine ca numar sau creste ca frecventa pe masura ce dezvoltarea leucemica progreseaza. Prin urmare, utilizarea combinata a acestor 2 markeri indica progresia tumorala prin reducerea semnalelor de fluorescenta generate de anticorpii anti-Sca1 si cresterea semnalelor de fluorescenta generate de anticorpii anti-Ly6C.
Figura 3:
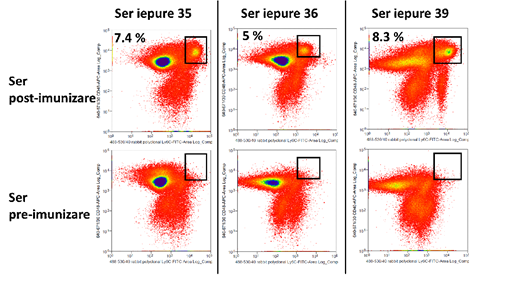
Dupa doua serii de imunizare, s-a testat reactivitatea serului fata de celulele hematopoietice ale soarecilor, prin citometria in flux. Imunizarea s-a facut pentru antigenul Ly6C, iar probele testate au provenit de la trei iepuri. Același tip de celule au fost marcate cu anticorp anti-iepure marcat FITC si cu ser (probe) sau fara ser (control de fundal), iar diagramele FACS au fost comparate pe baza intensitatii semnalului fluorescent si a procentelor de populatii pozitive. In cazul probelor tratate cu ser, legarea anticorpilor primari de suprafata celulara este evidentiata prin semnal FITC. Materialul biologic a fost reprezentat de celule de soarece extrase din maduva hematogena a animalelor. Rezultatele sunt prezentate in figura 4. In ansamblu, datele sugereaza ca serul post-imunizare prezinta anticorpi policlonali cu afinitate sporita in 66% din cazuri.
Figura 4:
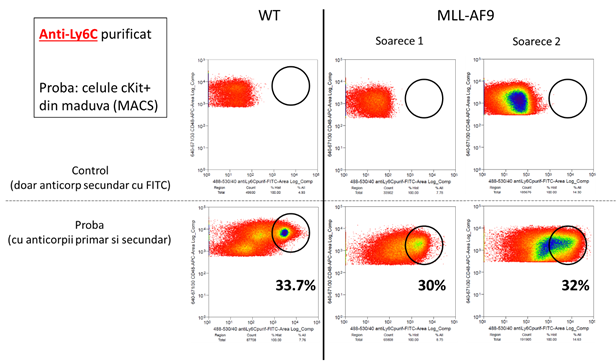
Dupa finalizarea etapelor de imunizare a iepurilor si sortarea limfocitelor producatoare de anticorpi, s-a primit de la parteneri o serie de fragmente imune scFV reactive la markerii antigenici Sca-1 si Ly6C. Analiza FACS (figura 5) indica faptul ca toate fragmentele sunt reactive fata de antigenii corespunzatori.
Figura 5:
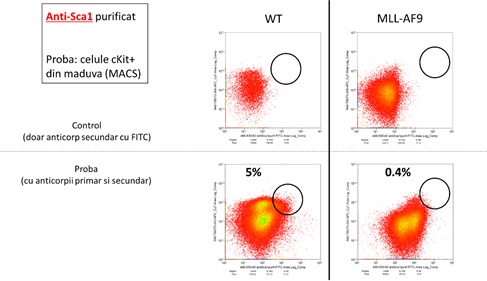
In concluzie, toate activitatile asumate de catre partenerul INCD-VB au fost indeplinite.
Publicatii si conferinte/congrese rezultate din aceasta finantare
